CINRYZE®
C1 esterase inhibitor (human)
Consumer Medicine Information
What is in this leaflet
Read all of this leaflet carefully before you start using CINRYZE. It provides information on:
What CINRYZE is used for
How CINRYZE is given
While you are using CINRYZE
Product description
Keep this leaflet. You may need to read it again. It does not contain all the information about CINRYZE.
If you have any further questions, ask your doctor or pharmacist.
All medicines have benefits and risks. Your doctor has weighed the benefits that CINRYZE will have for you against the risks.
This medicine has been prescribed for you. Do not pass it on to others. It may harm them, even if their symptoms are the same as yours.
If you get any side effects that worry you, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
If you have any concerns about using this medicine, ask your doctor. Follow your doctor’s advice even if it is different from what is in this leaflet.
The information in this leaflet is subject to change.
Please check with your doctor whether there is any new information about this medicine that you should know since you were last treated with this medicine.
What CINRYZE is used for
CINRYZE contains the human protein C1 esterase inhibitor as the active substance.
C1 esterase inhibitor is a naturally occurring human protein that is normally present in blood. If you have a low amount of C1 esterase inhibitor in your blood or your C1 esterase inhibitor is not working properly, this can lead to swelling attacks (called angioedema).
Symptoms of hereditary angioedema (HAE) may include:
stomach pains and
swelling of the:
hands and feet
face, eyelids, lips or tongue
voice-box (larynx), which may make breathing difficult
genitals
How CINRYZE works
In adults and adolescents, CINRYZE can raise the amount of C1 esterase inhibitor in blood and either prevent these swelling attacks from occurring or stop swelling attacks once they have begun.
Before you use CINRYZE
Do not use CINRYZE if you are allergic to:
C1 esterase inhibitor
or any of the other ingredients of CINRYZE (listed at the end of this leaflet)
It is important to tell your doctor if you think you have ever had an allergic reaction to any of the ingredients in CINRYZE.
Before you are given CINRYZE
Before you start treatment with CINRYZE, it is important that you tell your doctor if you have, or have had, problems with your blood clotting (thrombotic events). You will be carefully monitored if this is the case.
If you think you might be pregnant, are planning to get pregnant, or are breast feeding, ask your doctor for advice before using CINRYZE. There is limited information on the safety of CINRYZE use during pregnancy and breast feeding. Your doctor will discuss with you the risks and benefits of using this medicine.
Tell your doctor if you are on a controlled sodium diet.
This medicine contains sodium which should be taken into consideration.
If you have not told your doctor about any of the above, tell them before you are given CINRYZE.
If you begin to suffer from rashes, tightness of the chest, wheezing, or a fast heart beat once you have used CINRYZE then you should tell your doctor immediately.
When medicines are made from human blood or plasma, certain measures are put in place to prevent infections being passed on to patients.
These include careful selection of blood and plasma donors to make sure those at risk of carrying infections are excluded, and the testing of each donation and pools of plasma for signs of virus/infections. Manufacturers of these products also include steps in the processing of the blood or plasma that can inactivate or remove viruses. Despite these measures, when medicines prepared from human blood or plasma are administered, the possibility of passing on infection cannot be totally excluded. This also applies to any unknown or emerging viruses or other types of infections. The measures taken are considered effective for enveloped viruses such as human immunodeficiency virus (HIV), hepatitis B and hepatitis C viruses, and for the non-enveloped hepatitis A and parvovirus B19 viruses. Your doctor may recommend that you consider having vaccinations against hepatitis A and B if you regularly or repeatedly receive C1 esterase inhibitor products that have been taken from human plasma.
If you have any questions, talk to your doctor.
Taking other medicines
Always tell your doctor about other medicines you are taking, including those bought without a prescription.
Some medicines may affect the way other medicines work.
CINRYZE is injected so any food or drink you consume should not have any effect on CINRYZE.
How CINRYZE is given
A doctor or nurse may prepare and inject CINRYZE for you. Your doctor may also train you or a family member to prepare and inject CINRYZE. They will ensure you receive full instructions and training on how to use it.
If you do not understand the instructions ask your doctor or nurse.
How much is injected
For Adults and Adolescents
Treatment of swelling attacks
The contents from 2 vials of CINRYZE at the first sign of the onset of swelling.
The contents of 2 extra vials should be injected if the swelling is not under control after 60 minutes (more likely if the attacks are severe or if there are laryngeal attacks or if the start of treatment is delayed).
CINRYZE is administered into a vein at an injection rate of 1 mL per minute.
Prevention of swelling attacks
The contents from 2 vials of CINRYZE every 3 or 4 days for routine prevention against swelling attacks.
CINRYZE is administered into a vein at an injection rate of 1 mL per minute.
Prevention of swelling attacks prior to surgery
The contents from 2 vials of CINRYZE should be injected up to 24 hours before a medical, dental or surgical procedure.
CINRYZE is administered into a vein at an injection rate of 1 mL per minute.
For Children
Do not use CINRYZE in children before adolescence.
How to prepare CINRYZE
If your doctor considers that you should receive CINRYZE at home, the instructions below should be followed carefully.
Always wash your hands before doing the following steps. Try to keep everything clean and germ-free while you are preparing CINRYZE. Once you open the vials, you should finish preparing CINRYZE as soon as possible. This will help to keep them germ-free.
Use of a silicone-free syringe is recommended for reconstitution and administration of the product. For your convenience, a silicone-free syringe is provided in the Administration set.
Note: Two vials of CINRYZE are required for each dose.
You should prepare both vials according to steps 1 through 10.
1.Let the vials of CINRYZE and the vials of water reach room temperature.
2.Remove the plastic caps from the powder vial and the water vial to show the centre part of the rubber stopper.
Do not remove the rubber stopper.
3.Release the powder vial label by tearing down the perforated strip indicated by the inverted triangle.
4.Wipe the rubber stopper of each vial with an alcohol wipe or swab, and allow it to dry. Do not blow on the stopper to dry it. Place each vial on a flat surface. After cleaning, do not touch the rubber stopper with your hand or allow it to touch any surface.
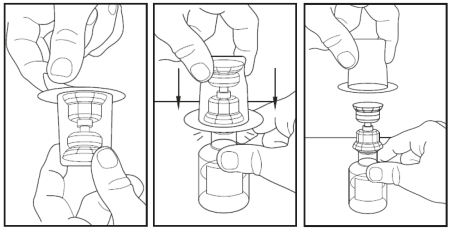
5.Note: the transfer device must be attached to the water vial before being attached to the powder vial.
Remove the protective covering from the top of the transfer device package.
Do not remove the transfer device from the package.
Place the water vial on a flat surface, and place the blue end of the transfer device over it, pushing down until the spike goes into the rubber stopper and the transfer device snaps in place.
The transfer device must be positioned completely upright before penetrating the rubber stopper.
Remove the plastic package on the transfer device and discard it.
Take care not to touch the exposed end of the transfer device.
6.Place the powder vial on a flat surface.
Turn the vial containing water upside-down and insert the clear end of the transfer device into the powder vial, pushing down until the spike goes into the stopper and the device snaps in place.
The transfer device must be positioned completely upright before penetrating the stopper. The water will be sucked into the powder vial.
If this does not happen, do not use the product.
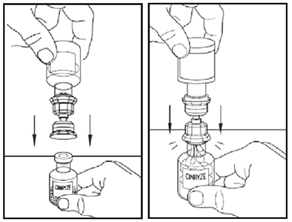
7.Once all the water is in the powder vial, gently swirl (do not shake) the powder vial until all the powder is dissolved.
Disconnect the water vial by turning it anti-clockwise. Do not remove the clear end of the transfer device from the powder vial.
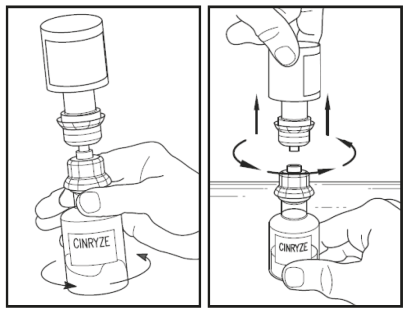
Look at the final solution before using it to make sure that CINRYZE is completely dissolved. Once dissolved, the solution in the CINRYZE vial should be colourless to slightly blue and clear.
Do not use the product if the solution is cloudy, discoloured or contains any particles. Throw it away and prepare a new vial of CINRYZE.
One vial of dissolved CINRYZE contains 5 mL of C1 inhibitor solution (500 IU). Prepare two vials of CINRYZE to make one dose of 10 mL (1000 IU). Repeat steps 1 to 7 using a new transfer device. Do not reuse the transfer device.
8.Using a sterile, disposable 10 mL syringe, draw back the plunger to allow approximately 5 mL of air into the syringe.
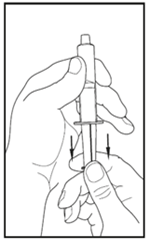
9.Attach the syringe to the clear end of the transfer device by turning it clockwise.
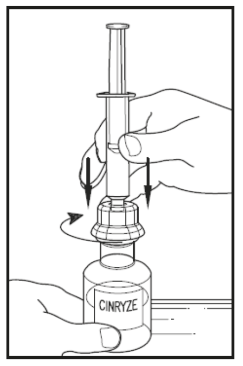
10.Gently turn the vial of CINRYZE upside down and inject air from the syringe into the vial.
Slowly pull as much of the dissolved CINRYZE as possible into the syringe. While holding the vial upside down, disconnect the syringe from the vial by turning it anti-clockwise and releasing it from the transfer device.
Remove any air bubbles by gently tapping the syringe with your finger and slowly pushing the air out of the syringe.
11.Using the same syringe, repeat steps 8 to 10 above with a second vial of CINRYZE to make one complete dose of 10 mL.
12.Inspect the reconstituted CINRYZE solution for particulate matter prior to administration; do not use if particles are observed.
Dispose of the vials with the transfer device attached to them.
CINRYZE should be administered within 3 hours after preparation. The dissolved CINRYZE solution may be stored at room temperature prior to injection. If not used within 3 hours after preparation, throw away the CINRYZE solution.
How to inject CINRYZE
Your doctor or nurse will teach you how to safely inject CINRYZE.
It is important that CINRYZE is injected directly into a vein and not injected into surrounding tissues and not injected into an artery.
Once you learn how to self-administer, you can follow the instructions in this leaflet.
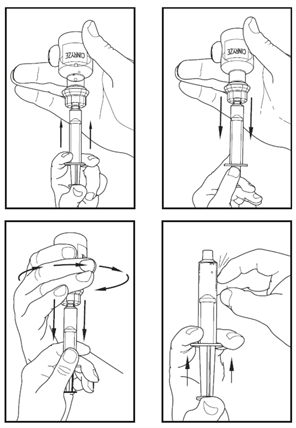
1.Attach a needle or venipuncture set to the syringe containing the dissolved CINRYZE solution. Fill the tubing with dissolved CINRYZE by gently pushing the plunger of the syringe.
Be careful not to spill the dissolved CINRYZE. This process replaces the air in the tubing with dissolved CINRYZE.
2.Apply a tourniquet and prepare the injection site by wiping the skin well with an alcohol swab.
3.As instructed by your doctor or nurse:
Insert the needle of the venipuncture set tubing into your vein.
Remove the tourniquet.
Make sure that the needle is in a vein.
Inject the dissolved CINRYZE product slowly over ten minutes (approximately 1 mL/min).
4.After infusing CINRYZE, remove the venipuncture set and discard. Cover infusion site with a plaster.
The small amount of CINRYZE left in the infusion set will not affect your treatment. Dispose of all unused solution, the empty vials, and the used needles and syringe in an appropriate container used for throwing away waste that might hurt others if not handled properly.
If too much CINRYZE is given (overdose)
No case of overdose with CINRYZE has been reported.
If you suspect that you have used more than you should have, contact your doctor or nurse immediately.
While you are using CINRYZE
Things you must do
If you notice signs or symptoms of a serious allergy or anaphylaxis (see Side Effects) while you are being given CINRYZE tell your doctor immediately as the administration of CINRYZE should be stopped immediately.
Use this medicine in one patient on one occasion only.
Things you must not do
Do not give or share your medicine with anyone else, even if they have the same condition as you.
Side effects
Tell your doctor as soon as possible if you do not feel well while you are being given CINRYZE.
Like all medicines, CINRYZE can cause side effects, although not everybody gets them.
Tell your doctor or pharmacist if you notice any of the following symptoms and they worry you:
reactions where the injection is given – symptoms include: pain, skin redness, bruising, discomfort, swelling, bleeding, itching, hardening of the skin, tingling, warmth or rash
nausea
vomiting
headache
dizziness
fever
hot flush
itching, flaking of the skin
rash
The above list includes the more common side effects of your medicine.
Contact your doctor or go to the Accident and Emergency at the nearest hospital immediately if any of the following occurs:
allergic reaction which may result in shortness of breath; wheezing or difficulty breathing; swelling of the face, lips, tongue or other parts of the body; or rash, itching or hives on the skin
pain and/or swelling of an arm or leg with warmth over the affected area
discolouration of an arm or leg
unexplained shortness of breath
chest pain or discomfort that worsens on deep breathing
unexplained rapid heart rate
numbness or weakness on one side of the body.
Although they are rare, the symptoms can be severe.
If you get any side effects that worry you (even side effects not listed in this leaflet), talk to your doctor or pharmacist.
After using CINRYZE
Storage of CINRYZE
CINRYZE powder in the vial has a shelf life of two (2) years when stored below 25°C (room temperature).
Do not freeze. Store in the original package in order to protect from light.
Do not mix CINRYZE with other materials.
Do not use CINRYZE after the expiry date which is stated on the carton or vials after “EXP”.
Once reconstituted, CINRYZE solution should be used immediately. If immediate use is not possible, the reconstituted product may be stored at room temperature and used within 3 hours.
Disposal of CINRYZE
If your doctor tells you to stop using CINRYZE or the pack has expired, ask them what to do if you have a pack left over.
Product description
What CINRYZE looks like
CINRYZE comes in a pack which contains:
CINRYZE vial of colourless glass, containing freeze-dried powder. The vial is sealed with a rubber stopper, and an aluminium seal with a plastic flip-off cap. There are 2 powder vials.
The solvent (water for injections to dissolve the CINRYZE powder) vial of colourless glass, which is closed with a rubber stopper and an aluminium seal with a plastic flip-off cap. There are 2 solvent vials.
Each pack also contains an administration set consisting of:
2 filter transfer devices
1 disposable 10 mL syringe
1 venipuncture set
1 protective mat.
Use of a silicone-free syringe is recommended for reconstitution and administration of the product. For your convenience, a silicone-free syringe is provided in the pack.
What CINRYZE contains
CINRYZE contains C1 esterase inhibitor, obtained from human plasma, as the active ingredient.
It also contains the following excipients:
sodium chloride
sucrose
sodium citrate dihydrate
valine
alanine
threonine.
Sponsor
Shire Australia Pty Limited
Level 39, 225 George Street
Sydney, NSW 2000
Australia
Telephone: 1800 012 612
This leaflet was prepared in April 2019.
Australian Registration Number:
AUST R 177513
CINRYZE is a trademark or registered trademark of ViroPharma Biologics, Inc.
SHIRE and the Shire Logo are trademarks or registered trademarks of Shire Pharmaceutical Holdings Ireland Limited or its affiliates.






